Introduction
The Rare Earth Elements (REEs) are the metal lanthanides La through Lu, the “f” block of the periodic table with atomic numbers 57 through 71, and the elements yttrium and scandium. REE’s unique luminescent, magnetic and catalytic properties make them essential to the United States economy in diverse applications ranging from catalysis to luminescent materials in displays. The US supply chain for these elements is tenuous, as China currently dominates their mining and refinement. The development of efficient, green technologies which can separate lanthanides, along with increased domestic mining of these elements, are essential for the rebuilding the domestic supply chain.
Our Research
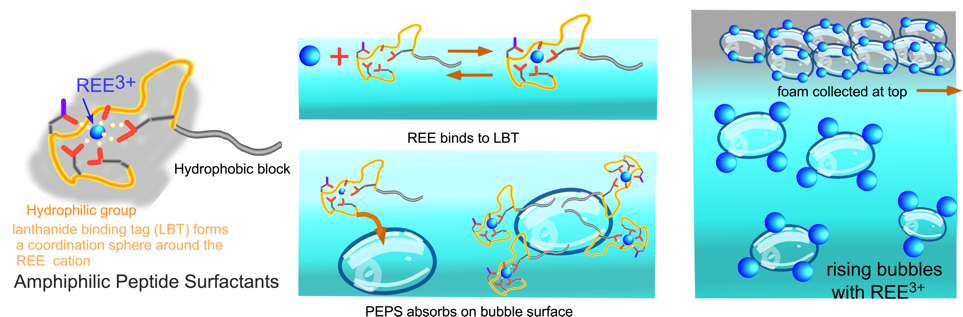
We are developing a bioinspired, green, ion foam flotation process for the selective recovery of rare earth elements from aqueous mining feedstocks in which the REEs are present in mixtures as cations in a +3 oxidation state. In ion foam flotation, a surfactant of opposite charge to the ion being recovered is added to the mixture, the surfactant binds to the ion and adsorbs to gas bubbles sparged through the solution. The bubbles are collected at the top of the solution as a foam to complete the recovery of the ions. Here we design surfactants in which the polar group is a lanthanide binding tag (LBT), a peptide containing a binding loop (comprised of a sequence of predominately hydrophilic amino acids) which forms a coordinating sphere of ligands around the cation in the cavity of the loop. These loops are present in natural proteins where they are designed to bind divalent calcium cations, and are re-adapted here to bind the trivalent rare earth cations. By changing the peptide sequence of the loops, the affinity of the loop to a particular REE cation can be achieved. A hydrophobic sequence of amino acids is added to one end of the hydrophilic loop to make the peptide amphiphilic, driving adsorption of these peptides, bound with REEs to bubble surfaces for foam recovery.